If you are looking at this as part of a wider course, please choose the right course to return to! (you will need to re-enter the password given out on the course)

Influenza & COVID-19

Influenza
Every year we eagerly await the 'flu letter' so we know what we are doing in the coming season.
The 26/27 flu letter arrived 26/2/26.
We began the 25/26 vaccine clinics from 1st Sept for the kids and pregnant ladies, and 1st Oct for the adult and clinical risk groups 18+. 25/26 and 24/25 are the only years we have staggered the starts and this is due to quicker waning immunity in older adults than in youngsters. The rationale is that an extra month to wait means the vaccine protection will carry through to the end of flu season. 26/27 remains the same.
It's a massive undertaking to deliver the flu programme.
As of 23rd October 2025 we had vaccinated over 10.4 million (10,436,395) people against flu and almost three million (2,987,313) people against COVID. By 8th Jan 2026 over 18.6 million eligible people were vaccinated.
UKHSA published modelling estimates which suggested that flu vaccination had prevented approximately 100,000 hospitalisations in the 24/25 season in England.
BUT vaccine uptake across all groups has consistently dropped since lockdown times.
Sadly, we hit the 25/26 flu season five weeks earlier than usual. Australia experienced a severe winter; France and Japan also reported very high activity. There was a mutated A strain in the summer which is always a risk when deciding on vaccine strains so far in advance. Hence, 25/26 was a VERY challenging season.
Antigenic shift and drift can be a real problem (see video below).... Fortunately the 25/26 vaccines maintained their effectiveness. PHEW!
COVID-19 has changed a lot too. If you want to REALLY geek out try this website...
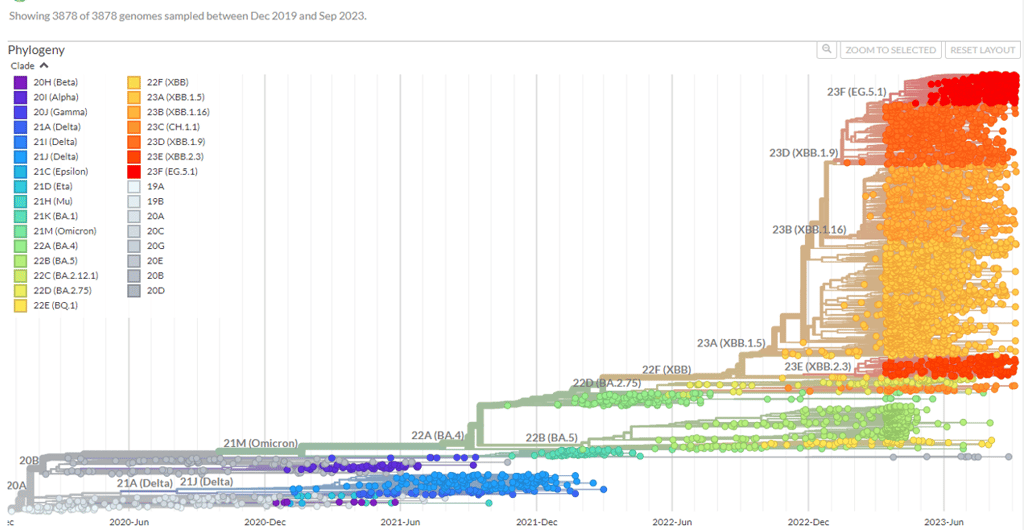
If you are particularly keen on the ins and outs of virus changes, also check out the section on SARS COV-2 epidemiology from the Green Book
Information on new variants under investigation is included in the weekly National flu and COVID-19 surveillance reports (if you are interested in this kind of thing!)
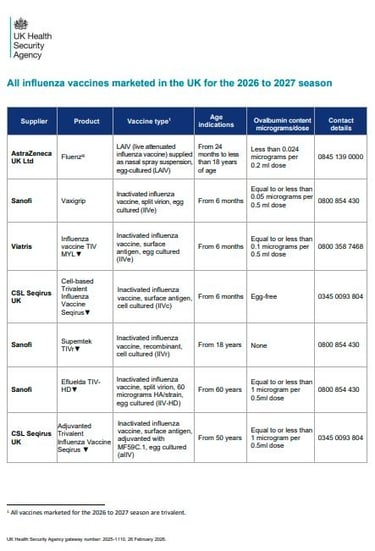
But back to flu. Let's take a closer look at the flu vaccines...

Ovalbumin content
LESS THAN 0.12MCG/ML = SAFE TO VACCINATE
(equivalent to <0.06mcg for 0.5ml dose)
Porcine Gelatine Content
Fluenz (nasal flu- LAIV) contains a small amount of highly purified porcine gelatine. This vaccine is the most effective option for children aged 2 and older but there is the (LESS effective) injectable alternative if required.
So, think carefully about how these conversations are had.... here's a blog with some practical tips for you.
Other queries about vaccine contents might be around microchips, dogs, sharks and moths...
Quick tip: Note the little black triangles... When providing patients with details of the vaccine, it is good practice to give them details of the brand and batch number. This will allow patients and carers to more accurately report suspected ADRs to the Yellow Card scheme.
Quick bit of history: if you are new to this, you may still hear flu vaccines being referred to as 'QIV' (quadrivalent influenza vaccine) or 'TIV' (trivalent influenza vaccines) ... more seasoned flu vaccinators have had to change their terminology a lot over the years, so if a 'TIV' slips in instead of an 'IIV' on occasion - you know what it means!! The changes to terminology were about future-proofing consistency of language to avoid confusion down the line. Makes sense, but some us us feel that IIV doesn't quite roll off the tongue the same as TIV or QIV... politics eh? Learn more about what is meant by 'valency' in the video below.

Want to know more about vaccine production?
Sanofi (the first company to make a flu vaccine) made a video...
Watch it later if you fancy it.
QUICK CHANGES-FROM-LAST-YEAR NEWS! (if this is not your first rodeo)
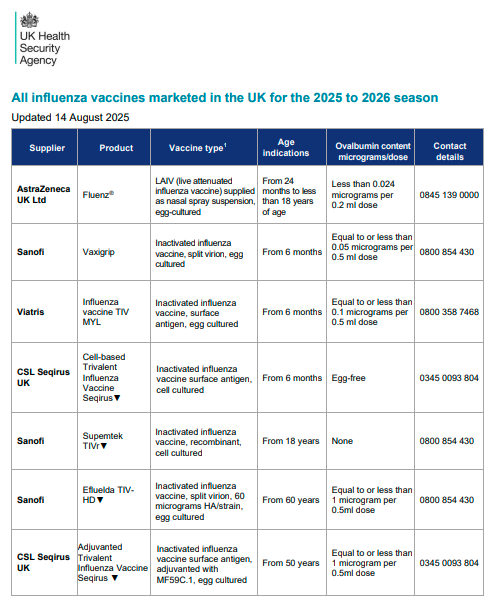
Previous years have seen quadrivalent vaccines (4 strains) but 25/26 said goodbye to the Yamagata B strain (due to not circulating for a while) and all UK vaccines are now trivalent.
In 25/26 aIIV became licenced from 50 years
IIVr made a return in 25/26!
LAIV now includes PGD provision for up to 25y in SEN schools.
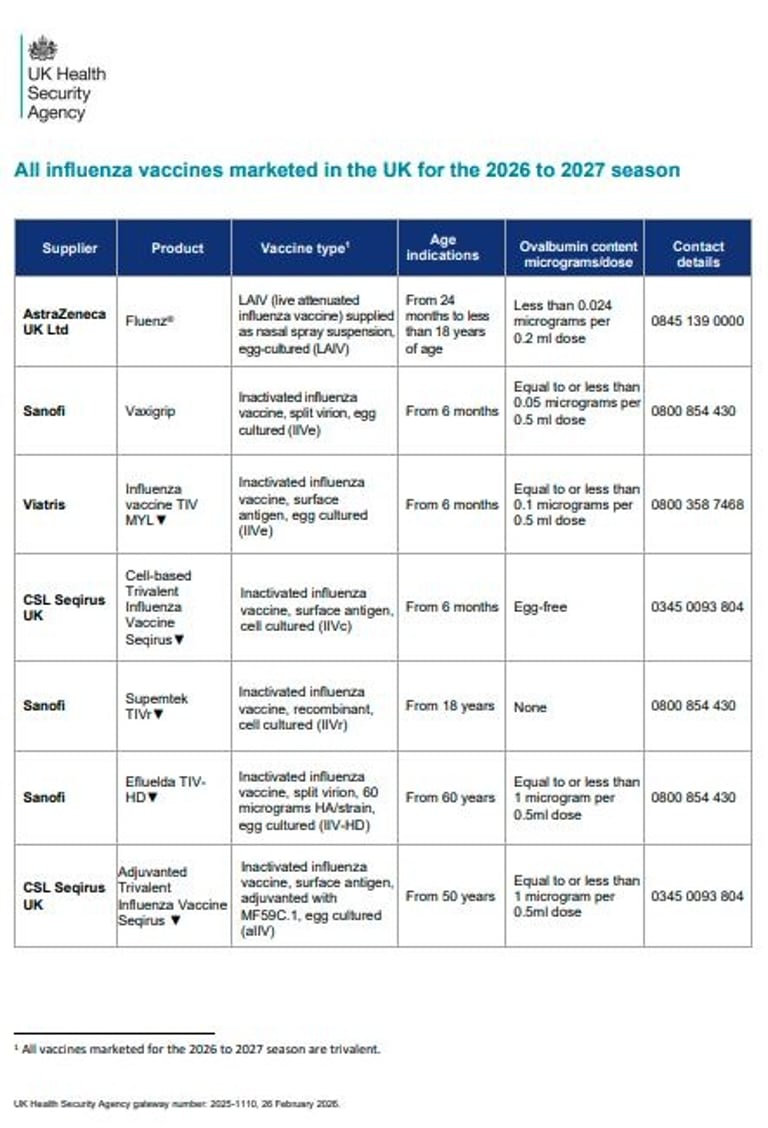
There are no changes to eligible cohorts for the 26/27 programme and the timing remains the same staggered (for some) start as last year.
Community pharmacy got involved with child flu vaccinations in 25/26. NHSE is currently undertaking a service evaluation of the delivery of this, where flu vaccines were administered to two to three-year-olds in community pharmacies during the 2025/26 campaign. JCVI also advise prioritising 2/3 year olds for 26/27.
There are some firm words in the flu letter about planning to do more for uptake in 26/27, especially in under-served groups. The new GP contract aims to address these inequalities (and QOF) more fairly.
NHSE has now published the GP additional guidance for seasonal vaccinations 2026/27. This provides information on recording COVID-19 and influenza vaccination events and payments, including where practices are collaborating in their PCNs. In previous years this guidance has only covered the influenza programme. For 2026/27 it has been expanded to cover both the COVID-19 and influenza programmes.
There have been 2 changes to the JCVI advice for adult flu vaccines for 26/27. Firstly, in those aged 65 years and older, IIVc should be considered equivalent to allV, llV-HD, and IIVr. And secondly, in those aged 50 years to 59 years in clinical risk groups, IIV-HD can be used off-label (60+ licence).
Looking at the vaccines table, IIVe seems to be very much taking a back seat now.
NEW legal mechanisms! Goodbye to National Protocols and hello to the VGD!


• LAIV is different from other flu vaccines – it is a live attenuated nasal vaccine and must not be injected
• Do not attempt to attach a needle
• Fluenz can be administered at the same time as, or at any interval from other vaccines including live vaccines
• Patient should breathe normally - no need to actively inhale or sniff
• The vaccine is rapidly absorbed so no need to repeat either half of dose if patient sneezes, blows their nose or their nose drips following administration
Administration of Nasal Flu Vaccine

IM administration
Note she doesn’t actually insert the needle in this demo (don’t forget to put it in!!!)


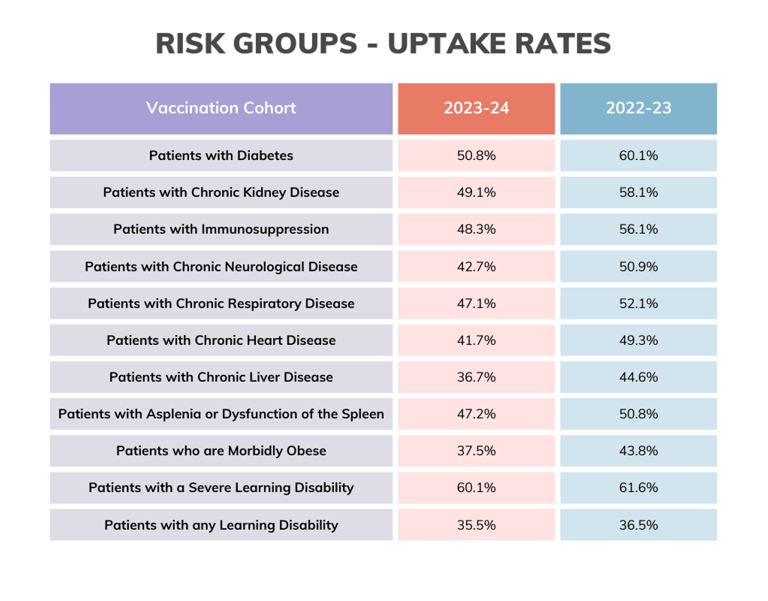
Results: "Access was NOT the primary issue underlying suboptimal vaccine uptake among participants in clinical risk groups, who instead cited low-risk perceptions of influenza infection and deficits of information about the relevance of vaccination for their condition management. Healthcare providers in non-primary care settings rarely discussed or recommended influenza vaccination across patient pathways, despite being able to address the concerns raised by participants in clinical risk groups." (I.E. vaccine uptake is not just about putting on the late night and weekend clinics! People may well make more efforts to show up if they knew WHY they should)


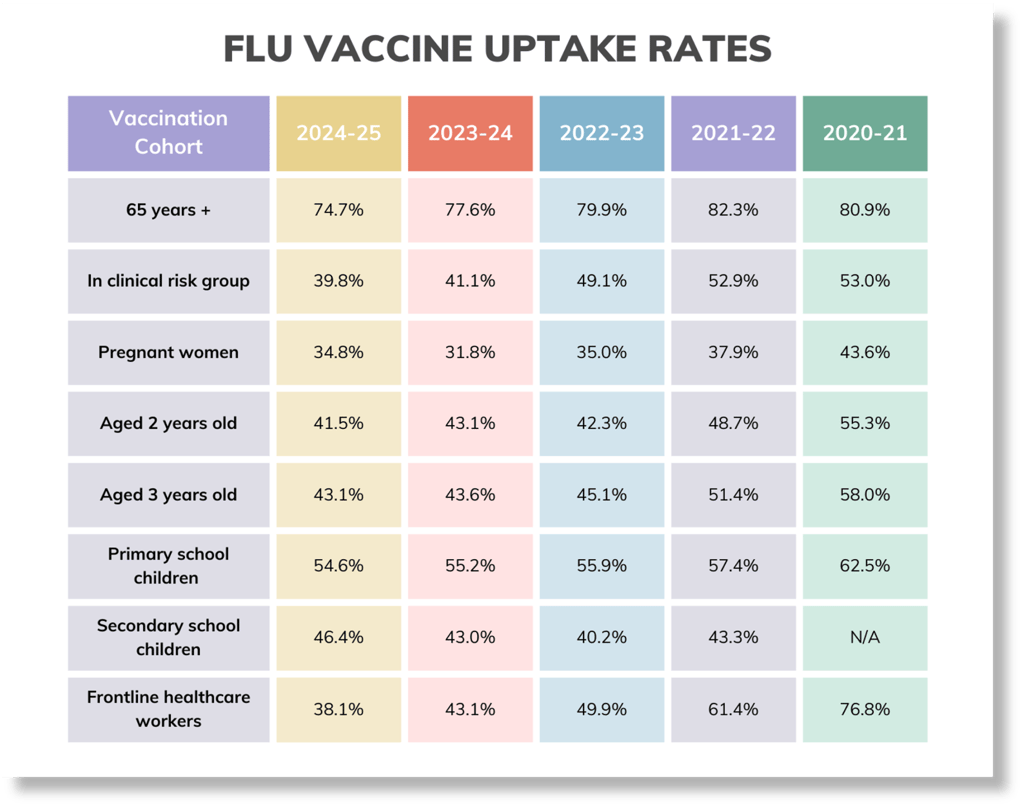
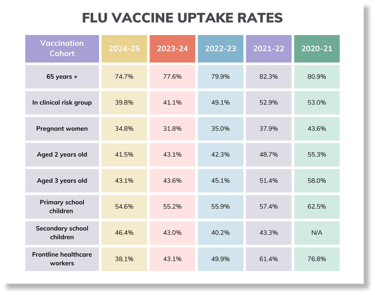
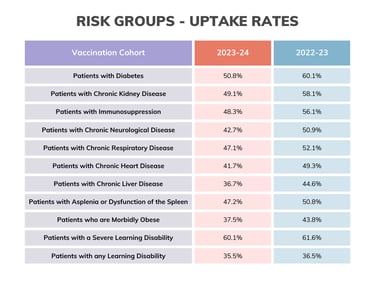
Here's a quick vaccine uptake snapshot as of data published 26th March:
Check out the GP patients uptake here and frontline healthcare workers here.
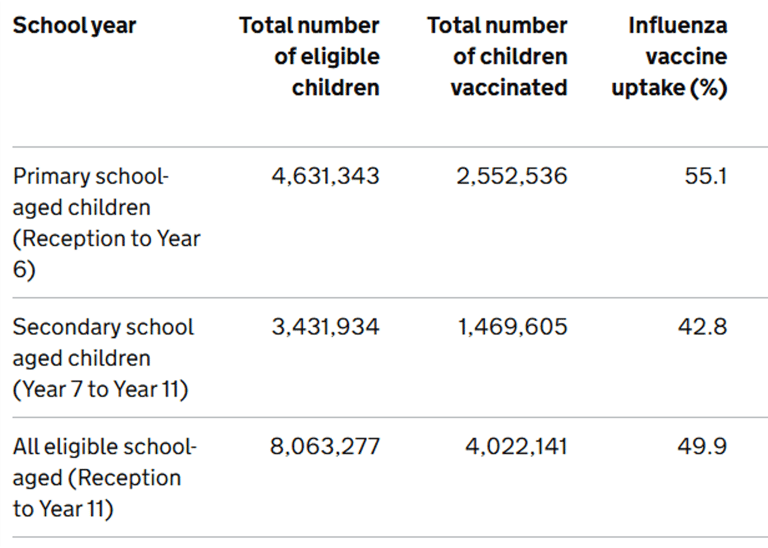
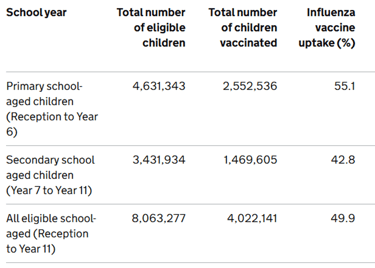
Summary is so far: 65+: 74.5%, clinical risk groups: 40.6%, pregnant women, 38.4%: 2 yr olds: 43.5%, 3yr olds: 44.8%, frontline HCPs (us): 46.1% (better than the 37.5% compared to last year). For children, the reception years are doing the best at 56.3%, with the age 2's and year 11's achieving the lowest uptake at 43.4%.
Flu vaccination in adult social care settings
Within the 99.1% of older adult care home providers who had updated their seasonal vaccination data as of 16 February 2026:
11.3% of total staff are known to have received a flu vaccination for the 2025 to 2026 season
66.7% of total residents are known to have received a flu vaccination for the 2025 to 2026 season
Within the 99.0% of younger adult care home providers who had updated their seasonal vaccination data as of 16 February 2026:
9.1% of total staff are known to have received a flu vaccination for the 2025 to 2026 season
54.5% of total residents are known to have received a flu vaccination for the 2025 to 2026 season
Reflection points (for ALL routine vaccines):
Do you know your local uptake figures?
In YOUR clinic, which are the most difficult groups to reach? Why?
Do you see similar results/patterns to the national figures?
What other vaccines might these groups be missing out on?
Do YOU have yours?
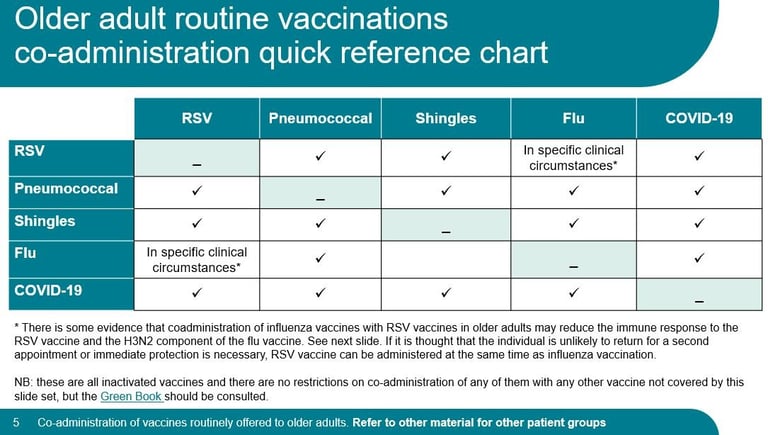
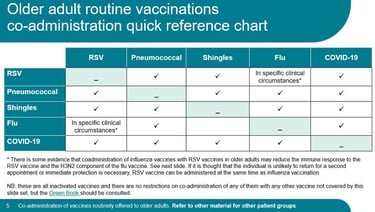
Giving more vaccines?
Click here for a useful UKHSA slideset about co-administration of adult vaccines from April 2026 (scroll down about a third of page to the heading 'Routine immunisation schedules')

COVID-19
At least 15–20 million lives saved globally in the first year alone—and many more since—according to the modelling.
COVID-19 vaccination is currently offered to eligible individuals twice a year, in Autumn and Spring campaigns.
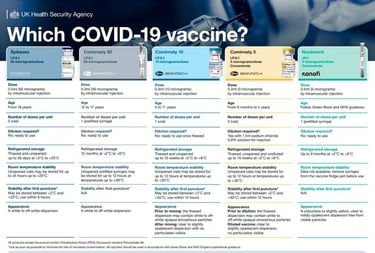
The Autumn 2025 booster programme was 1st Oct-31st Jan. Spring vaccinations started on the 13th April and runs to June - targeting high-risk groups only. Check out the latest variant vaccines (and the helpful poster). Plenty of ongoing research (boosters & new variants). Hundreds of vaccines in development.
Vaccines evolving – bivalent options, new variants, nasal options? Combined vaccines? Where is the 2 in 1 COVID and flu jab up to? Could this be with us in 2026?
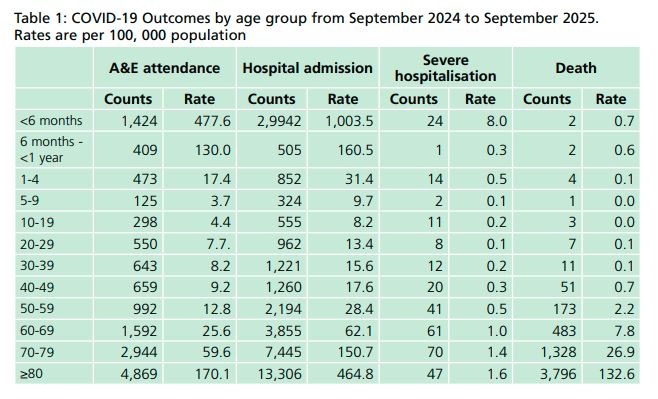
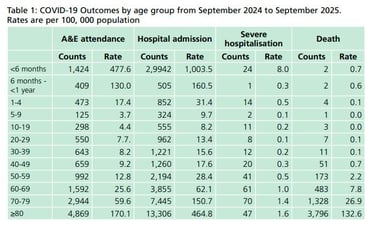
The primary aim of the national COVID-19 vaccination programme remains the prevention of severe illness (hospitalisations and deaths) arising from COVID-19, but, as the currently available COVID-19 vaccines provide only limited protection against mild and asymptomatic disease, the focus of the programme is now on offering vaccination to a more limited cohort who continue to be most vulnerable and is most likely to directly benefit from vaccination. 80% of deaths have been in those aged 70 years and over (check out the table below) and most of the excess all-cause mortality consisted of older age groups with more than 90% of excess deaths in those aged over 75 years.
Eligible individuals are offered a single dose of vaccine during the campaign, provided at least three months have passed since their last dose. Severely immunosuppressed individuals may be eligible for COVID-19 vaccination between campaigns. (JCVI considers that eligible children aged six months to four years of age should continue to receive two primary doses of vaccine). Could COVID-19 move to an all year round prog? JCVI minutes from June 2025 indicate it's up for consideration.
Green Book Chapter has now been updated and simplified (March 18th 2026) ready for the spring boosters. Eligibility is now 75+, care home residents, and IC 6m+.
ELfHC module updated too! Check it out for further important details if you are delivering COVID-19 vaccines. PGD and VGD are also Spring-ready.
Find campaign resources HERE and HERE.
Legal changes have lead to some confusion and misunderstandings with other vaccines - check your legal mechanisms! Also note: NEW legal mechanisms! Goodbye to National Protocols and hello to the VGD!
Pandemic disruption has left much catching up to do with other vaccines

Useful videos for further learning!